Vaccine Technologies
What is West Nile Virus?
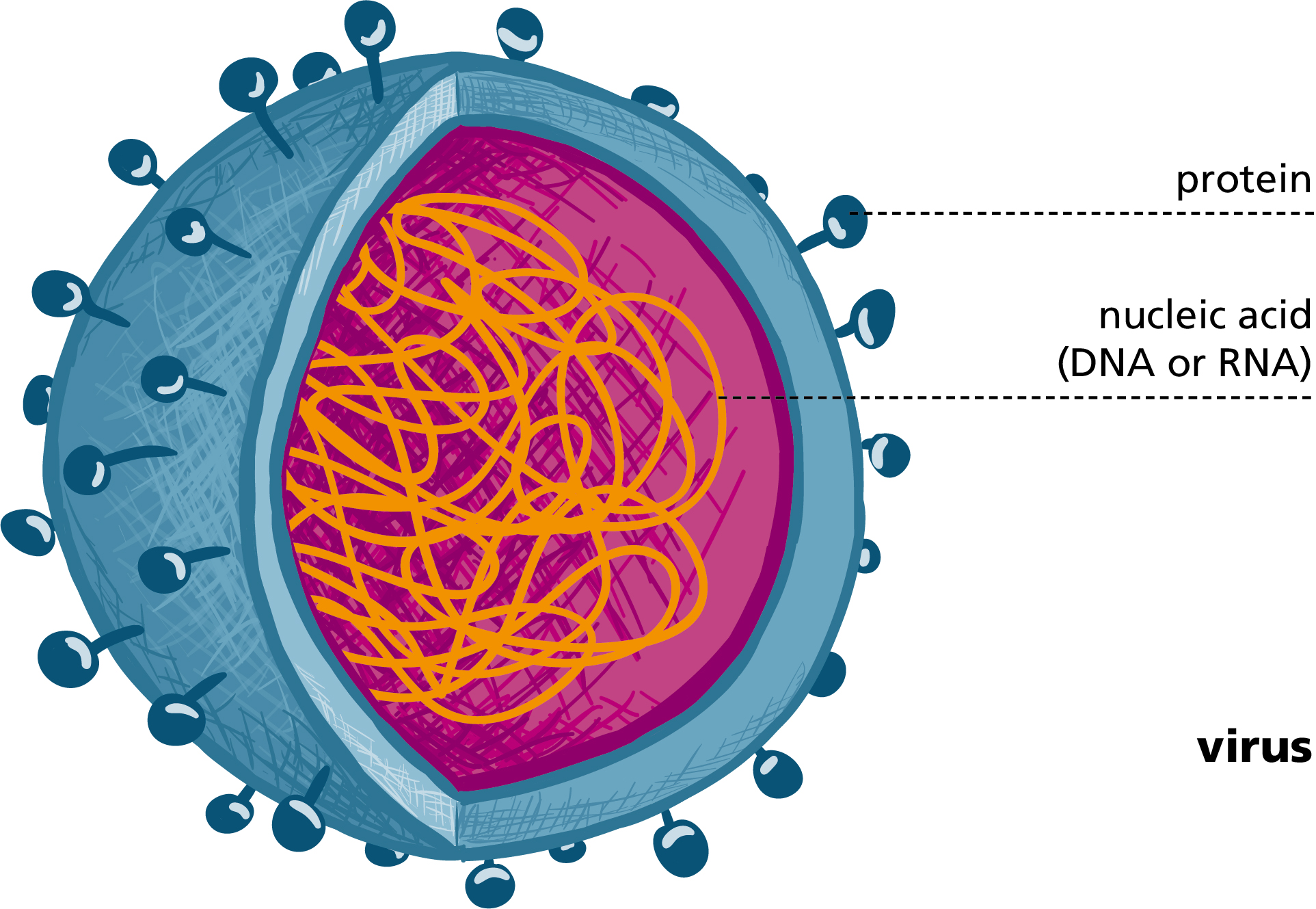
West Nile Virus (WNV) is an enveloped RNA virus belonging to the family of flaviviruses.
Other flaviviruses include pathogens such as dengue and yellow fever, Zika virus and tick-borne encephalitis.
The virus was first detected in 1937 in the West Nile district of Uganda, which is where it gets its name.
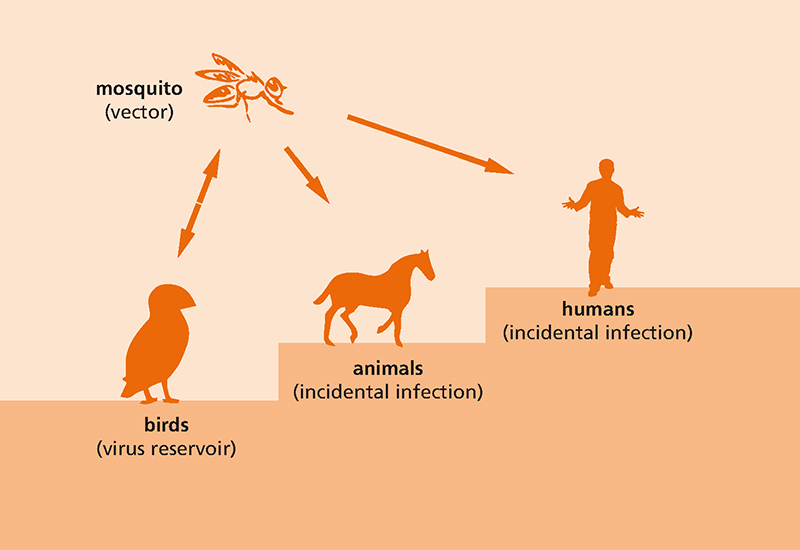
It mainly affects birds but is transmitted by various types of mosquito, which in turn leads to infections in mammals, e.g. horses and humans. The occurrence of the virus in birds and horses constitutes an animal epidemic that has to be disclosed to the authorities in Germany; infections in humans also have to be reported.
What symptoms are associated with WNV?
WNV is fatal for many types of birds. In humans, the infection usually proceeds without any symptoms or is similar to an influenza infection. Clinical courses can, however, also be more serious, accompanied by back pain, shivering fits and a rash. The virus affects the nervous system in around one per cent of those infected, which can lead to meningitis, paralysis and other neurological symptoms – in rare cases also death. WNV poses a greater risk to immunodeficient and older people in particular.
No antiviral therapy exists at present and symptoms are treated symptomatically.
Why is the virus now also spreading across Germany and Central Europe?
As the primary hosts of the virus, birds are an ideal taxi for spreading the virus far and wide due to their seasonal migratory movements. Unlike most other tropical pathogens, WNV is also transmitted by mosquitoes native to Central and Northern Europe, especially Culex mosquitoes.
The only thing curbing the spread of the disease is the climate. A constant temperature of over 20°C is required for the virus to sufficiently propagate within the mosquitoes and enable the infection to spread. As this was rarely the case in Central and Northern Europe in the past due to the region’s distinct seasons, the virus has not been able to spread here.
However, a shift is now being observed with the milder winters brought by climate change.
The European Centre for Disease Prevention and Control has been recording the rising spread of the virus in Europe and its neighboring countries for many years now.
In Germany, the virus was first detected in birds and horses in Saxony, Brandenburg, Mecklenburg-West Pomerania and Bavaria in 2018.
The first three infections in Germany (Saxony, Brandenburg and Berlin) were verified in 2019. However, it has to be assumed that there are up to one hundred times more undiagnosed infections: on the one hand because doctors in Germany are not yet expecting to have to deal with the virus and, on the other, because no symptoms have been reported.
Distribution of West Nile virus infections in humans in the EU/EEA countries and EU neighbouring countries 2010 to 2019
Status November 2019, data source: European Centre for Disease Prevention and Control. © ECDC
Vaccinations
Several vaccines have been approved for horses since 2005, which are now being increasingly administered in Germany.
A vaccine is yet to be approved for humans. Coordinated by Fraunhofer IZI, potent vaccine candidates were developed between 2011 and 2014 as part of the West Nile Shield Project funded by the EU. However, considerable expenditure is still required for their further clinical development. Until then, the only preventive strategy is to avoid mosquito bites.
Expertise and cooperation opportunities at Fraunhofer IZI
Headed up by Dr. Sebastian Ulbert, the Vaccine Technologies Unit develops diagnostic technologies and prevention strategies for infectious diseases, including WNV. Diagnostic tests have already been developed at the institute that are able to specifically analyze WNV and differentiate it from other flaviviruses. Furthermore, new vaccine candidates have been developed and preclinically tested that are now available for further clinical development.